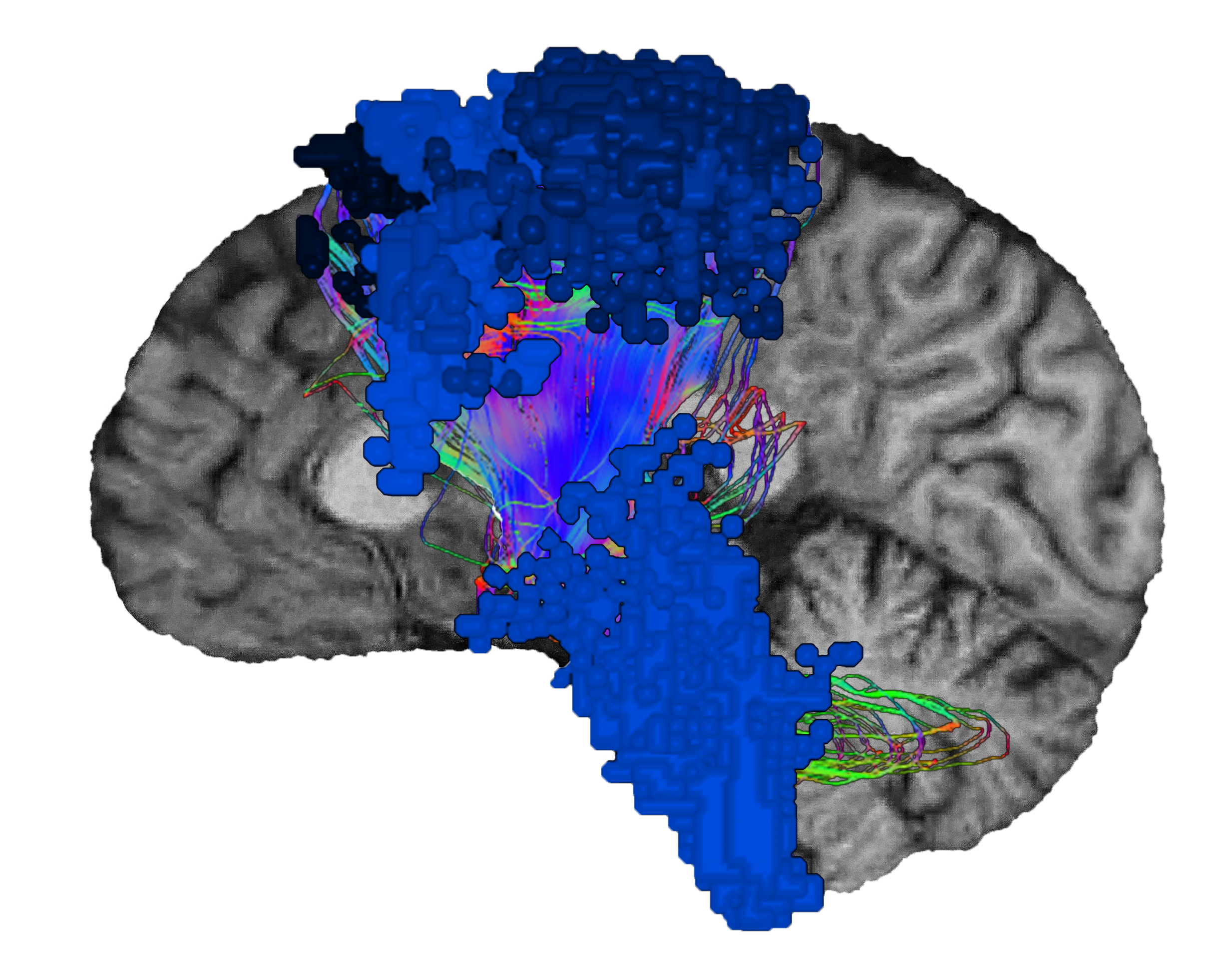
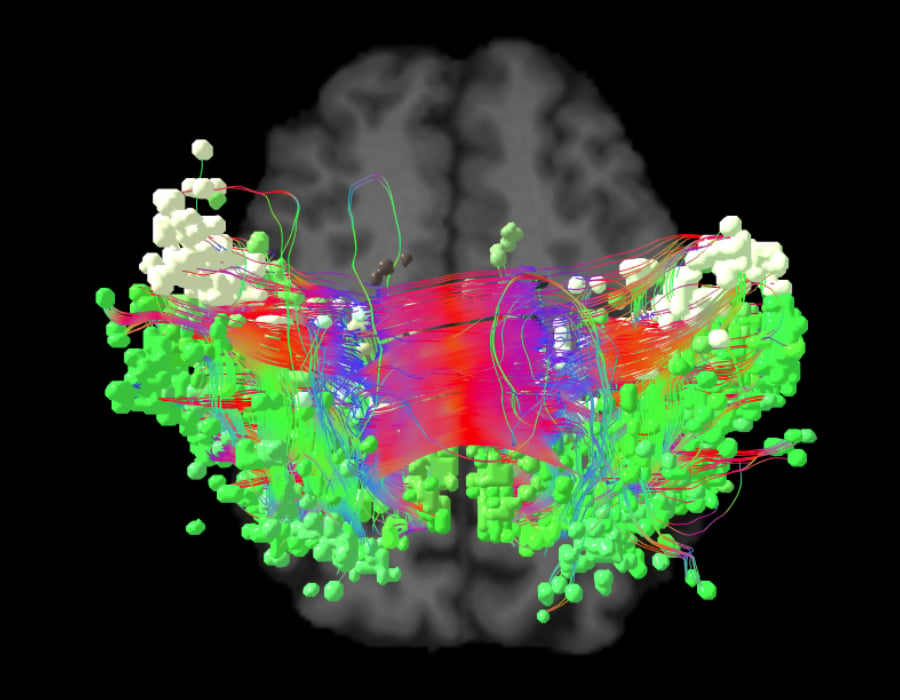
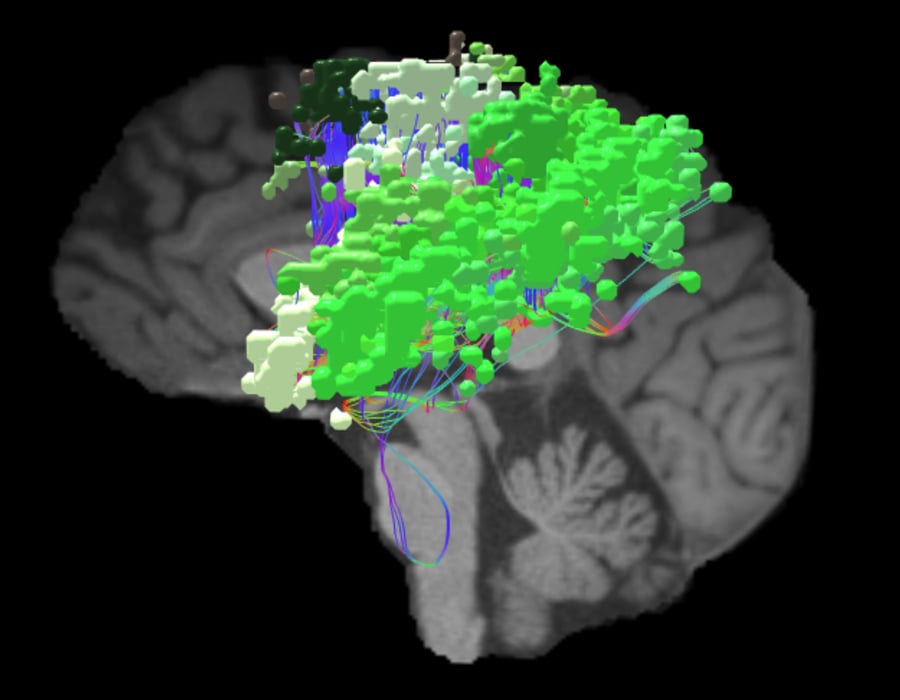
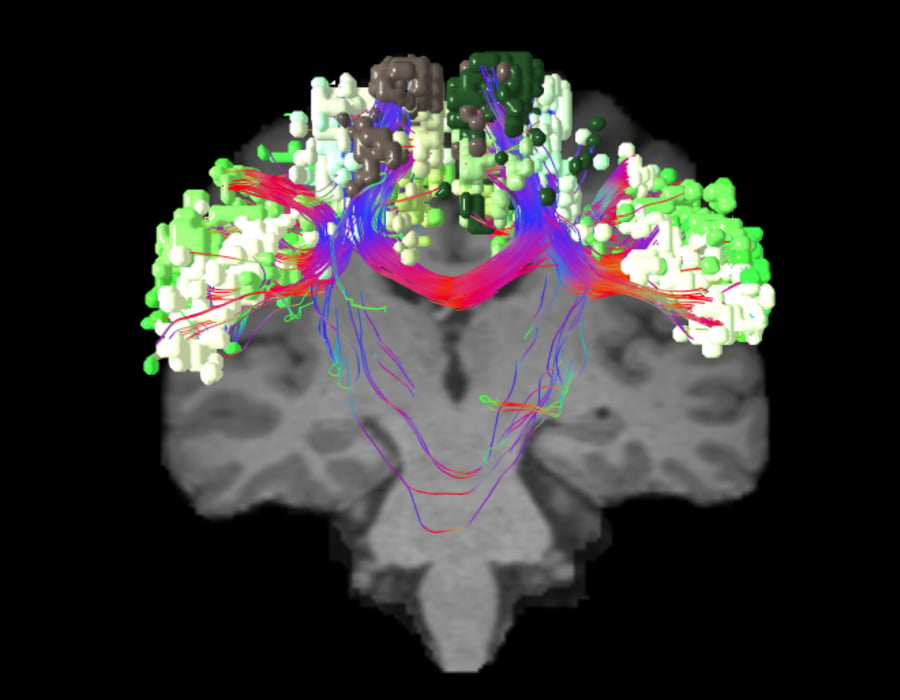
Neurologists have long been interested in how the brain gives humans the ability to move and physically function. The earliest discovery of the sensorimotor network came about in 1870 when German neurologist, Eduard Hitzig, and anatomist, Gustav Fritsch, studied motor function in dogs.5They applied electrical stimulation to certain parts of the dogs’ cortex6 and identified regions where direct stimulation caused contralateral limb movements.5
Scottish neurologist, David Ferrier, also studied the effects of stimulation on the brain, but he expanded the scope of the stimulation to evoke movements of body parts from different regions of the cortex,7including the eyelids, face, mouth, hands, and feet.
These studies and others paved the way for modern research, which has begun to evaluate the role the sensorimotor network plays in learning, skill acquisition, and memory.
The primary motor cortex, somatosensory cortex, auditory and visual cortices have been shown to demonstrate plasticity during learning exercises with behavior validation.8 In addition to learned tasks during early developmental stages,10 learning exercises can include processes relating to attention, expectancy, learning strategy, and motivation in response to a learned signal stimulus.8
As the phenomenon of neuroplasticity has shown, physical experiences can result in changes to the brain's functional connectivity.10 Motor learning exercises, such as skilled finger movements,11 can lead to cortical reorganization in areas of the sensorimotor network responsible for motor learning and areas responsible for the perceptual changes that occur during motor learning.10
In addition to motor learning during waking moments, movements occuring during active (REM) sleep are thought to maintain and improve the sensorimotor system.12 Active sleep may also provide opportunity for cortical repair after sustaining an injury or during pathological states.12
Furthermore, repetitive transcranial magnetic stimulation (rTMS) has been shown to induce changes in sensory perception and motor performance in key areas of the sensorimotor network.13 Using rTMS to improve sensorimotor capabilities offers the possibility to modulate sensory perception and motor performance, and it poses new and exciting opportunities for neuroscience13 and treatment.